

Adamantane Annulation to Arenes: A Strategy for Property Modulation of Aromatic π-Systems
Takaku Yoshihara, Hiroki Shudo, Akiko Yagi*, Kenichiro Itami*
J. Am. Chem. Soc.2023, ASAP. DOI: 10.1021/jacs.3c02788
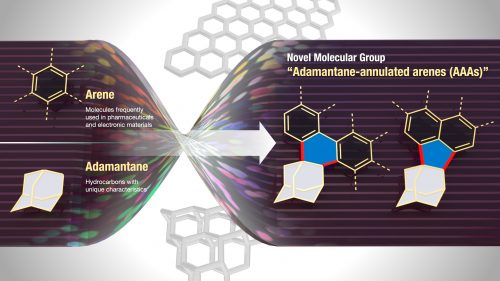
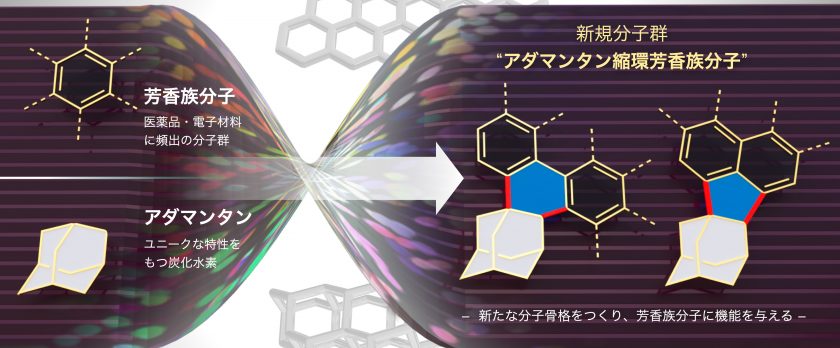
Peripheral structural modifications of arenes are widely used to control or improve the optoelectronic properties, molecular assembly, and stability of aromatic π-materials as well as to explore new functions. However, known modifications are often tedious and complex; therefore, a simple yet powerful modification strategy is needed. We discovered that annulation with a simple adamantane scaffold exerts a significant impact on the properties, alignment, and stability of aromatic π-systems. This unprecedented adamantane annulation was achieved by a two-step transformation of metallated arenes and 4-protoadamantanone, generating a range of adamantane-annulated arenes. Analysis of structural and electronic properties uncovered unique effects of the process, such as high solubility and enhanced conjugation. The oxidation of adamantane-annulated perylenes produced strikingly stable cationic species with emission extended to the near-infrared region. This simple property modulation of aromatic π-systems would not only create potentially ground-breaking π-materials but also novel nanocarbon materials, such as diamond–graphene hybrids.