

Yuuki Ishii, Sanae Matsuura, Yasutomo Segawa, and Kenichiro Itami
Org. Lett. 2014, ASAP, DOI: 10.1021/ol500643c
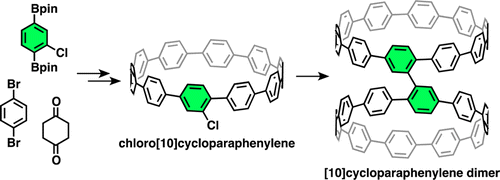
The first synthesis and dimerization of monochlorinated [10]cycloparaphenylene (chloro[10]CPP) are described. By assembling a chlorinated 1,4-diborylbenzene unit with brominated and borylated cis-1,4-diphenylcyclohexane units by Suzuki–Miyaura coupling, a triangle-shaped chloro-containing macrocycle was synthesized. The acid-mediated “cyclohexane-to-benzene” aromatization afforded chloro[10]CPP without losing the chloro group. By the action of Ni(0) complex, chloro[10]CPP was converted to a directly connected [10]CPP dimer, which would be an ideal precursor for the “carbon nanobelt”.