

Qianyan Zhang, Katsuaki Kawasumi, Yasutomo Segawa, Kenichiro Itami, Lawrence T. Scott
J. Am. Chem. Soc. 2012, ASAP. DOI: 10.1021/ja306992k
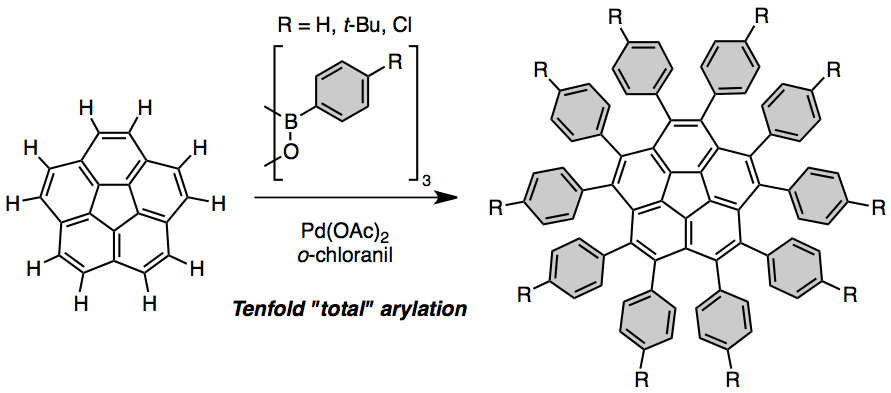
All ten C–H positions on the rim of corannulene can be arylated by repetitive palladium-catalyzed C–H activation. To relieve congestion among the ten tightly packed aryl substituents in the product, the central corannulene adopts a nearly planar geometry.