

Takao Fujikawa, Nobuhiko Mitoma, Atsushi Wakamiya, Akinori Saeki, Yasutomo Segawa, and Kenichiro Itami
Org. Biomol. Chem. 2017, Accepted Manuscript, DOI: 10.1039/C7OB00987A
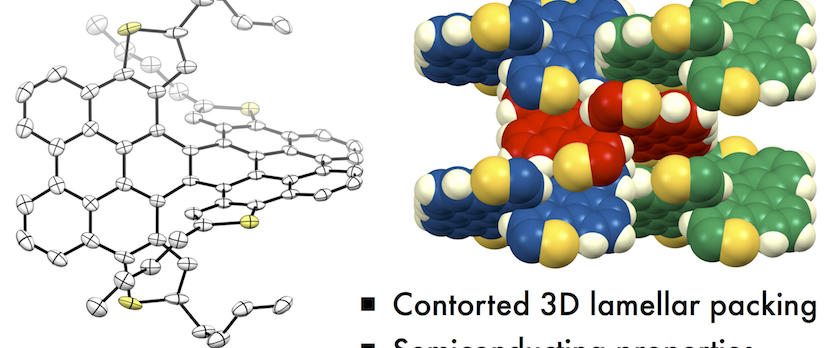
The synthesis and properties of a new π-extended double [6]helicene 2 and a dithia[6]helicene 3 are described. Compared to the previously reported parent double-helicene molecule 1, the introduction of n-butyl groups successfully improved the solubility, which allowed an experimental investigation into the electronic structure of 2 and 3 by photophysical measurements and cyclic voltammetry. The characteristic two-blade propeller structures of 2 and 3 were unambiguously determined by single-crystal X-ray diffraction analysis. The crystal packing structure of 2 exhibited a contorted two-dimensional stacking, whereby molecules of n-pentane were incorporated in the stacks. Despite the presence of n-butyl groups, 3 formed a unique three-dimensional stacking lattice in the crystal. Time-resolved microwave conductivity measurements revealed that the double helicenes (1–3) exhibited transient conductivities. An organic field-effect transistor fabricated using 3 was found to function as a p-type transistor.